A baby’s gut bacteria may predict future type 1 diabetes decades before diagnosis
Study identifies several potential bacterial biomarkers that could predict type 1 diabetes up to 20 years before the onset of the disease
Embargo 2301H UK time Saturday 25 March
The make-up and volume of bacteria in the gut, known as the microbiome, in 1-year-old infants may predict type 1 diabetes decades before any clinical signs of disease appear, suggests new research, published in Diabetologia (the journal of the European Association for the Study of Diabetes [EASD]).
Type 1 diabetes most often occurs in children and adolescents and is a disease in which the body’s own immune system gradually destroys the pancreatic islet beta cells that make insulin, for which early biomarkers prior to diagnosis are lacking.
While autoantibodies (biological markers of autoimmunity) are traditional markers for type 1 diabetes, they are rarely detected before the age of 6 months, and their peak incidence is between 9 and 36 months. Gut microbial biomarkers as early as 12 months could offer prediction opportunities and preventive strategies well before the onset of autoantibodies.
“Our findings indicate that the gut of infants who go on to develop type 1 diabetes is notably different from healthy babies and that several microbial biomarkers associated with future disease may be present as early as 1 year of age,” says co-lead author Dr Malin Belteky from Crown Princess Victoria’s Children’s Hospital, Sweden.
She adds, “This discovery could be used to help identity infants at highest risk of developing type 1 diabetes before or during the first stage of disease and could offer the opportunity to bolster a healthy gut microbiome to prevent the disease from becoming established.”
Type 1 diabetes is related not only to genetic predisposition, but also environmental factors such gut health and gut bacteria composition. The gut’s resident microbes are known to influence metabolism, body weight, the development of disease and the immune system.
Most previous studies investigating environmental risk factors have compared healthy children to those with an increased genetic risk for, or a family history of, type 1 diabetes, and found a reduced abundance of short-chain fatty acid-producing microbiota – bacteria and other microbes in the digestive system that help prevent inflammation – in children with early onset autoantibody development. But these studies are limited in their ability to evaluate other factors that can protect against or trigger an autoimmune response in the general population.
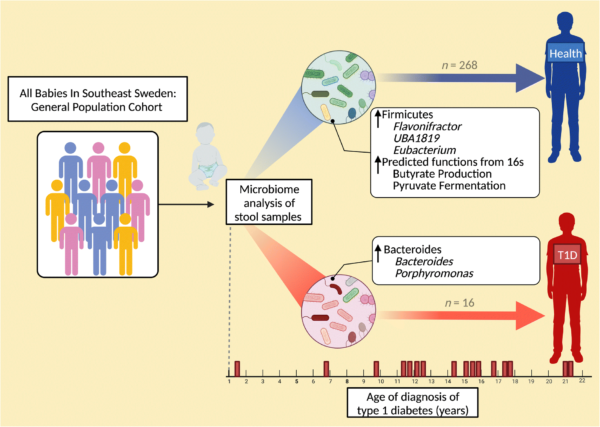
To find out more, researchers used data from children participating in the longitudinal, general population All Babies In Southeast Sweden (ABIS) study, to analyse gut microbiota taken from the stool samples of babies at around 1 year old.
In total, 16 babies who went on to develop type 1 diabetes at an average age of 13 years old (the youngest at 1 year and 4 months, and the oldest at 21 years and 4 months), and 32 control infants who remained healthy up to 20 years of age, were matched for geographical region, siblings at birth, residence type, duration of breastfeeding, and month of stool collection.
Parents completed questionnaires at the time of birth and at when the baby reached 1 year old, and also filled in a diary during the baby’s first year with information about pregnancy, nutrition, and lifestyle factors such as smoking and alcohol use.
The data were then analysed to determine which families and species of microbes were inhabiting the infant’s intestines and how the abundances of these microbes differed among samples.
The analyses found that core microbiome of healthy control infants versus those with a future diagnosis of type 1 diabetes were significantly different at one year of age, and that specific bacteria contributed to these differences in a large way.
Specifically, infants who went on to be diagnosed with type 1 diabetes had higher numbers of Firmicutes (Enterococcus, Gemella and Hungatella) and higher levels of Bacteroides (Bacteroides and Porphyromonas), that promote inflammation and are known to be involved in immune response.
And they also had far fewer species of health-promoting bacteria compared to healthy controls, including Firmicutes such as Anaerostipes, Flavonifractor, Ruminococcaceae UBA1819 and Eubacterium, that help maintain metabolic and immune health. These species produce an important short-chain fatty acid, butyrate, that helps prevent inflammation and fuels the cells of the gut lining, which keeps the colon healthy.
Overall, species that promote inflammation such as Alistipes (more abundant in healthy control infants) and Fusicatenibacter and Ruminococcus (mixed patterns of abundance between cases and controls) were the strongest factors for differentiating both control infants and those with future type 1 diabetes. The mixed pattern of total vs. relative abundance in key bacteria warrants further investigation, researchers say.
“Although the average age at which diabetes was diagnosed in our study was more than a decade after samples were collected, we identified distinct microbial signatures at 1 year of age and noted a decrease in butyrate-producing bacteria as seen in previous studies in high-risk populations,” says co-lead author Patricia L Milletich from the University of Florida, USA.
According to co-author Professor Eric Triplett from the University of Florida, USA, “The autoimmune processes usually begin long before any clinical signs of disease appear highlighting how differences in the make-up of the infant gut microbiome could shed important light on the complex interaction between the developing immune system, environmental exposures in childhood, and autoimmunity. Studies with much larger cohorts of prospectively-traced individuals will be required to establish which are the strongest biomarkers and how effectively they can predict disease.”
The authors note some limitations to their study, including that it is an observational study so can’t establish cause, and the relatively small sample size, added to which the gut microbiome varies widely among different populations, so the differences observed in this Swedish study might not be generalisable to infants elsewhere.
