Optimising indoor light conditions to mimic the natural light-dark cycle could help mitigate adverse metabolic effects of extended periods of exposure to indoor lighting
Embargo: 2301H UK time Wednesday 2 February
A new study published in Diabetologia (the journal of the European Association for the Study of Diabetes (EASD) finds that the timing of exposure to bright light can have a significant influence on postprandial (post-meal) glucose metabolism, thermoregulation, and energy expenditure during sleep in overweight, insulin-resistant adults.
The research was conducted by Jan-Frieder Harmsen, Patrick Schrauwen, and colleagues at the Department of Nutrition and Movement Sciences, NUTRIM School for Nutrition and Translational Research in Metabolism, Maastricht University Medical Center, Maastricht, The Netherlands. It aimed to determine whether redesigning indoor lighting to more closely mimic the natural light-dark cycle could help improve metabolic health. The authors say: “Optimising indoor light conditions to more closely mimic the natural light/dark cycle holds promise to prevent adverse effects on whole-body energy and glucose metabolism associated with the nowadays prevailing constant indoor lighting conditions.”
Light levels are known to be the most important time cue to synchronise the body’s circadian clock to the 24-hour day-night cycle. Previous research has shown that exposure to bright light during the evening or night influences glucose metabolism and is associated with being overweight or obese, as well as an increased risk of cardiovascular diseases.
In the modern world light exposure – including artificial light – is widespread and available around the clock, resulting in most people being exposed to light at home, in the workplace, or from the screens of electronic devices during the hours of darkness. The detrimental effects of light at night (LAN) include acutely elevated blood glucose and insulin levels after meals, and LAN exposure is associated with an increased incidence of type 2 diabetes mellitus (T2DM) in elderly people. The researchers note: “Our modern society also faces a lack of sufficient time spent under bright light conditions during daytime and in fact, most time is spent indoors under artificial lighting under much lower light levels compared to natural daylight outdoors.”
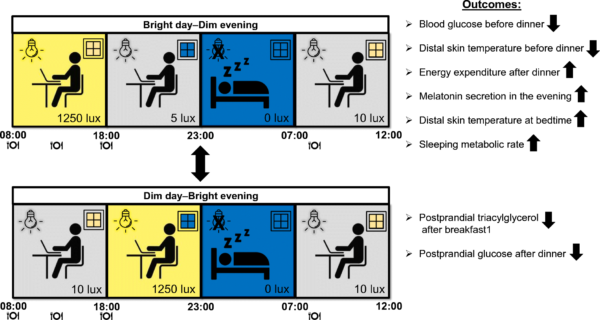
A mixed-sex group of 14 overweight (average BMI of 29.6kg/m2, on the borderline between overweight and obese), insulin-resistant volunteers between the ages of 40 and 75 were recruited for the study which was split into two sessions. Subjects were all non-smokers, had an average age of 67 years, and were in general good health. Participants stayed in a respiration chamber for the whole of each session, which lasted 40-hours from 18:00h on day 1 to 12:00h on day 3 and included two overnight sleep periods. A respiration chamber enabled the measurement of oxygen consumption and carbon dioxide production, allowing the calculation of total energy expenditure and sleeping metabolic rate (SMR).
One session (Bright day – Dim evening) comprised a period of bright light during daylight hours (08:00-18:00h), followed by dim light during the evening (18:00-23:00h), while the situation was reversed in the other Dim day – Bright evening session. Participants were in darkness during the night from 23:00h to 07:00h in both sessions, and meals were provided at 08:00h for breakfast; 13:00h for lunch; and 18:00h for dinner.
The quantity of food provided for each subject was determined by their daily energy requirement, based on their SMR which was obtained during their first night in the chamber. The macronutrient composition of the food (% protein, carbohydrate, and fat) was kept constant between sessions, as was the relative caloric value of each meal with breakfast accounting for a quarter of the day’s energy, while lunch and dinner represented 30% and 45%, respectively. Blood samples were taken before breakfast and dinner, then at 30-minute intervals in the 4 hours after both meals to determine levels of insulin, melatonin, triglycerides (fats), and glucose in both fasted and post-meal conditions.
The study found that spending the day in bright light (08:00-18:00h) led to lower blood glucose levels in the run-up to dinner compared spending the day in dim light. This Bright day – Dim evening pattern also had a beneficial effect on energy expenditure by maintaining a high SMR and increasing the body’s energy use during dinner.
In contrast, exposure to the Dim day – Bright evening condition was found to cause a reduction in SMR and energy expenditure in the period after dinner. This illumination cycle also heavily suppressed the release of the hormone melatonin, levels of which should rise 2-4 hours before bedtime in response to reduced natural light levels, and which plays an important role in regulating sleep cycles. Since even small changes in energy expenditure can contribute to the development of obesity and metabolic diseases in the long-term, the authors note that more longitudinal research is needed to investigate how light exposure influences regulation of body weight and metabolism.
To the surprise of the authors, postprandial glucose levels following dinner were reduced in the Dim day – Bright evening sequence. This reduction occurred despite insulin levels being unchanged, and previous research on this subject has demonstrated that the change in blood sugar could be the result of reduced gut activity and lower carbohydrate absorption. The team suggest that combining bright light during the day and dim light in the evening may promote digestion of food and could help explain why individuals exposed to light in this way exhibited higher after-dinner blood glucose than when the conditions were reversed.
It was also found that the Bright day – Dim evening condition supported a more diurnal variation in skin temperature: with a more pronounced reduction during daytime known to be mediated via vasoconstriction of blood vessels through the sympathetic nervous system, and with a transient increase in skin temperature as soon as the lights were dimmed at 18:00h indicative of vasodilation through parasympathetic nervous activity. The Dim day – Bright evening sequence lowered skin temperature less during daytime and led to a further lowering after increasing brightness levels at 18:00h, which is likely to postpone the onset of sleep.
Although not directly measured in this study, decreased skin temperature through vasoconstriction during daytime is indicative of higher blood pressure, while increased skin temperature through vasodilation in the evening is indicative of lower blood pressure. This diurnal fluctuation in blood pressure is desirable for cardiovascular health and the dip in blood pressure during the night is even used as a positive prognostic marker among medical experts. Therefore, the authors point out: “The combination of bright light during daytime and dim light during the evening seems promising to improve variation in blood pressure in a population that is prone to develop cardiovascular complications in the long-term.”
The authors conclude: “These findings suggest that the indoor light environment of insulin-resistant individuals can modulate metabolic parameters in a time-of-day dependent manner to influence metabolic health in the long-term…In the future, more research is required to exploit different light regimes in office buildings and home settings in their potential to prevent metabolic diseases.”
All News
